Mission & Objectives
Advancing fundamental understanding of kisspeptin‑centered hypothalamic mechanisms linking metabolism to pubertal control, with a focus on early‑onset obesity.
Our Mission
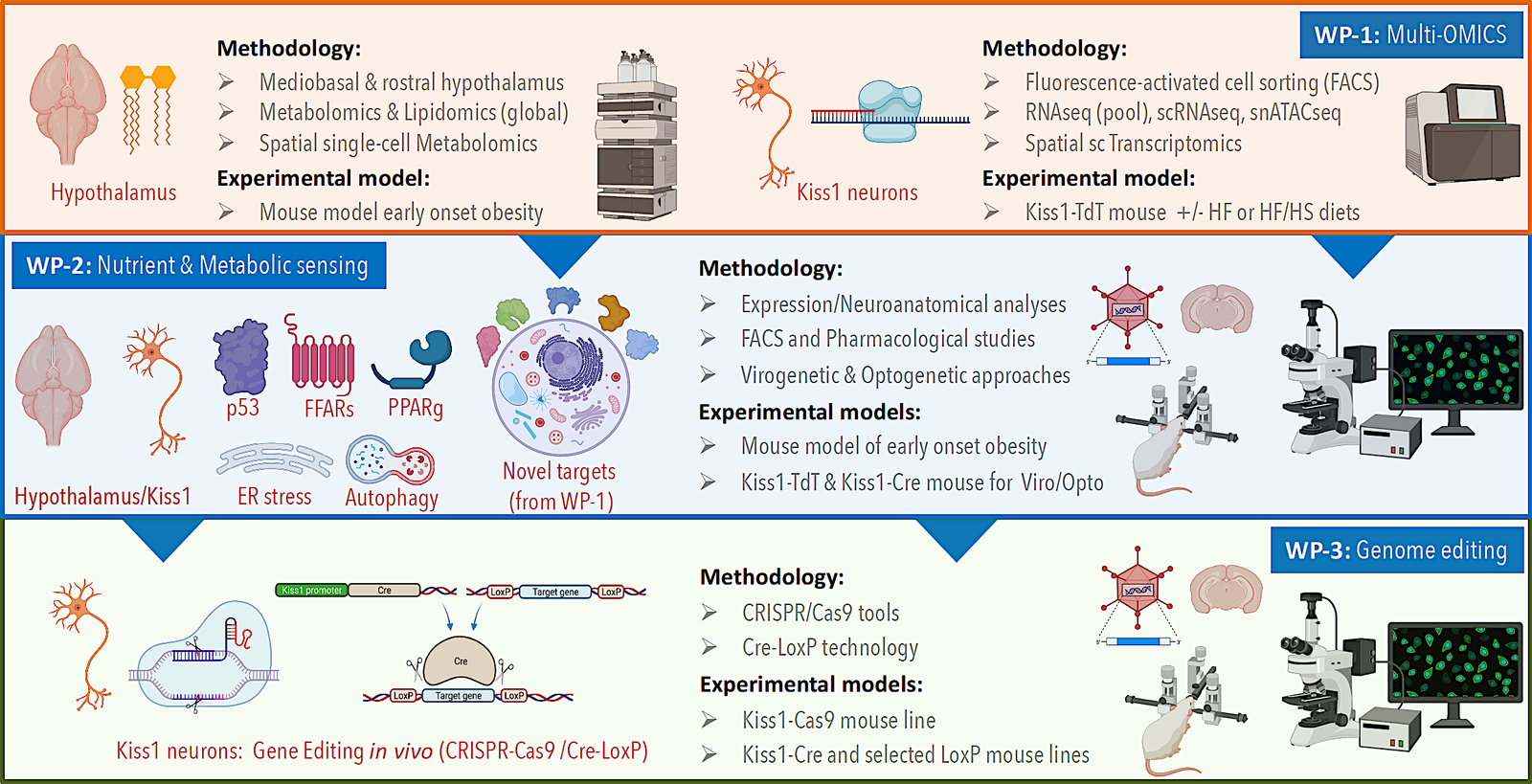
DOPA‑Kiss aims to generate a comprehensive framework for how hypothalamic Kiss1 neurons decode metabolic and hormonal signals to regulate the onset of puberty, why early‑onset obesity perturbs this process, and how sex differences shape these mechanisms; the program integrates multi‑omics mapping, studies of lipid and nutrient‑sensing pathways (e.g., FFAR/PPAR/TGR5; AMPK/mTOR/SIRT1), and in vivo functional genomics (Cre/LoxP and CRISPR) to establish causal mechanisms and translational relevance.
Scientific Rationale
Childhood and early‑onset obesity are rising concerns with significant impact on pubertal timing and long‑term health, pointing to hypothalamic mechanisms that integrate energetic and hormonal cues.
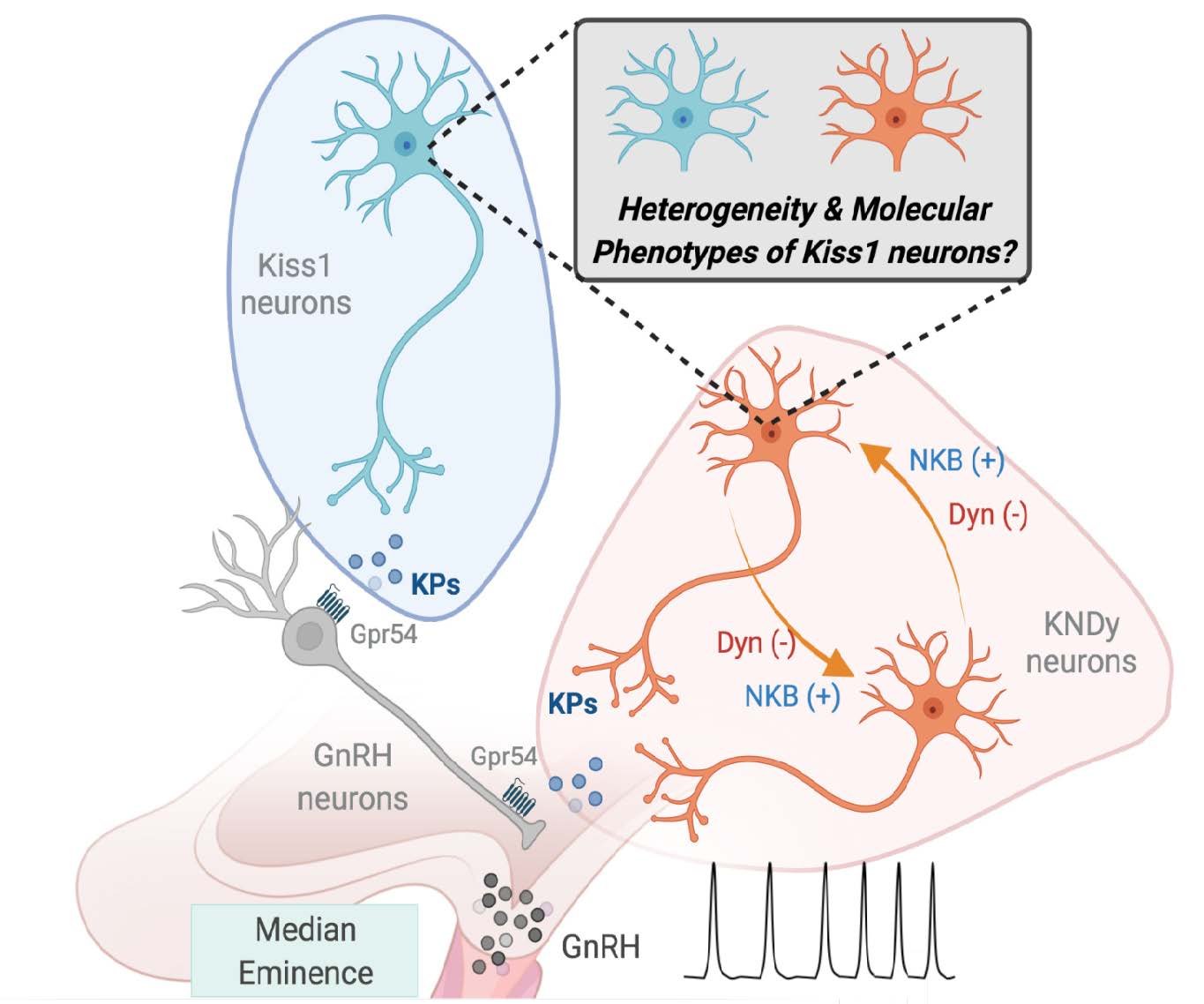
Kiss1 neurons are central nodes in the brain control of puberty, yet how metabolic information is decoded within Kiss1 circuits to advance or disrupt pubertal progression remains incompletely defined.
DOPA‑Kiss addresses this gap by combining systems‑level molecular profiling with targeted in vivo perturbations to define pathways that link metabolic status to pubertal control and its alterations in obesity.
Core Objectives
The project is structured around three interlinked objectives:

Characterisation of neuronal diversity
Define the molecular diversity, developmental dynamics, and functional signatures of Kiss1 neuronal populations via integrative multi‑omics across pubertal maturation and early‑onset obesity.
Elucidation of metabolic modulation
Map lipid and nutrient‑sensing mechanisms impacting Kiss1 function and hypothalamic circuits, including FFAR/PPAR/TGR5‑related pathways and cellular energy sensors (AMPK/mTOR/SIRT1).

Experimental validation of causal relationships
Apply Cre/LoxP and CRISPR‑based genome editing in vivo to test candidate pathways in Kiss1 neurons, quantify effects on pubertal timing and reproductive axis, and assess male–female differences.
Approach
DOPA-Kiss employs a multidisciplinary methodology that combines:
✅ Spatial and single‑cell multi‑omics to map hypothalamic and Kiss1‑specific molecular landscapes across pubertal development and early‑onset obesity.
✅ In vivo functional manipulations through Cre/LoxP and CRISPR/Cas to establish causal roles of prioritized pathways in Kiss1 circuits.
✅ Nutrient‑sensing studies to identify metabolite‑driven determinants of pubertal regulation, including lipid‑sensing routes with translational potential.
✅ Comparative analyses across sexes and models to maximize robustness and clinical relevance.
By integrating these approaches, the project will bridge the gap between fundamental neuroendocrine research and the development of novel strategies to address puberty-related disorders.