Main Achievements
Key scientific advances and milestones achieved during the DOPA-Kiss project.Overview
Since inception, DOPA‑Kiss has delivered advances clarifying how hypothalamic circuits translate metabolic cues into pubertal control, with an emphasis on early‑onset obesity and sex differences. Combining integrative multi‑omics, nutrient‑sensing studies, and in vivo genome editing, the project is generating causal insights with translational relevance.
Highlights of Scientific Progress
Molecular characterisation of neuronal diversity
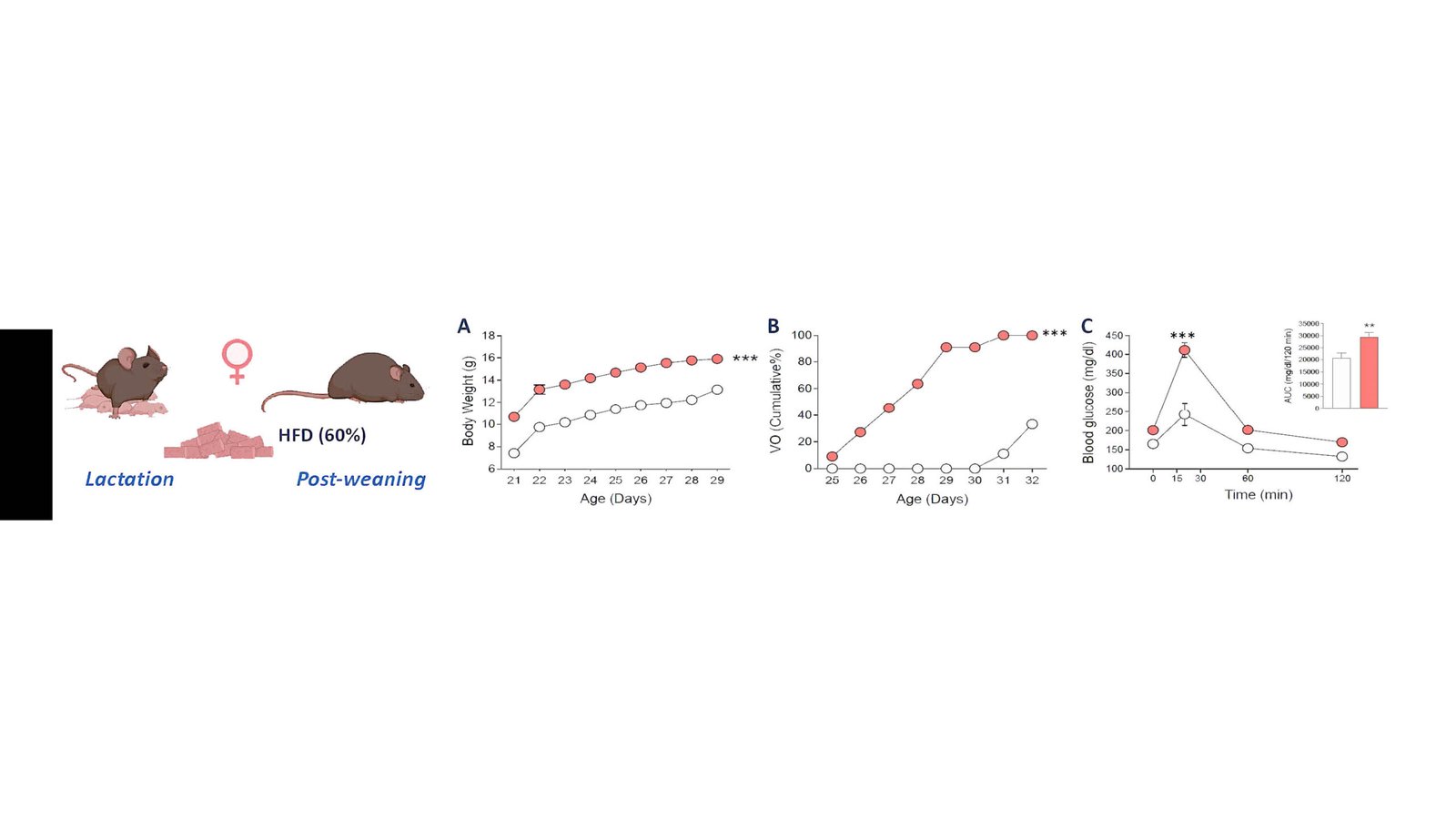
First hypothalamic lipidomic mapping across female pubertal maturation and early obesity, highlighting candidate lipid‑sensing routes for pubertal regulation.
Mechanisms of metabolic modulation
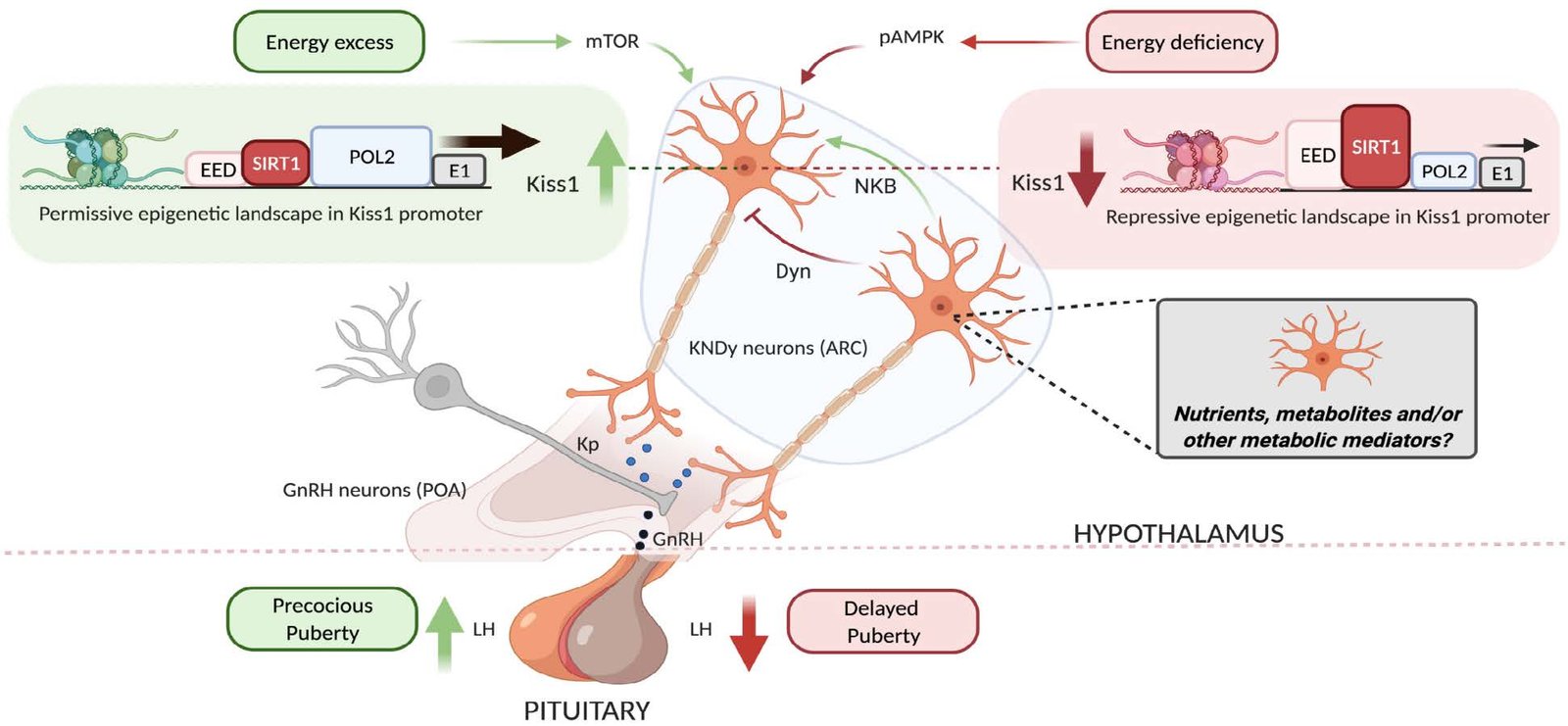
Authoritative review synthesizing neuronal circuits controlling puberty and mechanisms for its metabolic regulation, integrating recent advances from the project.
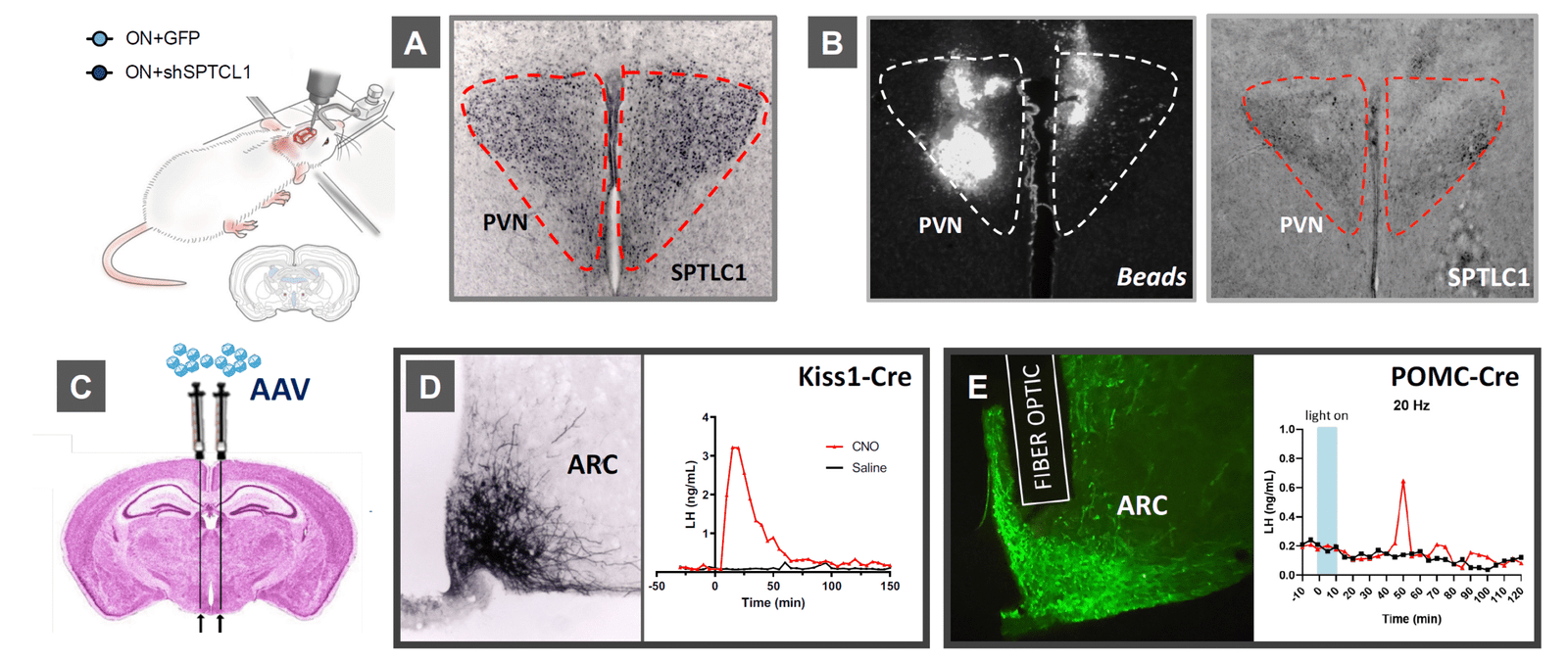
Genome editing to establish causality
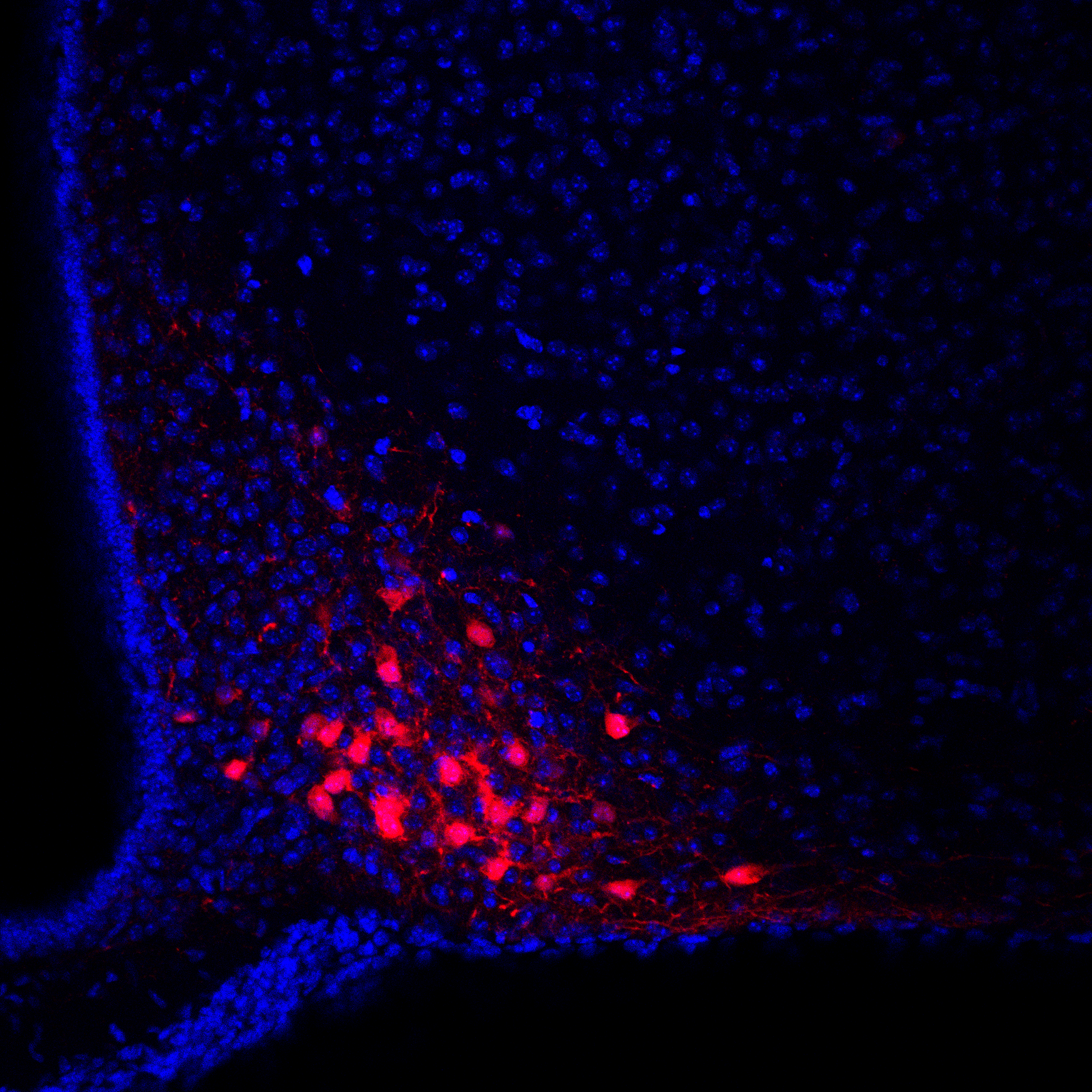
Astrocytic kisspeptin signaling modulates the reproductive axis, supporting non‑neuronal mechanisms relevant to obesity‑associated pubertal changes and targets for in vivo validation.
Publications and Dissemination
Results from DOPA-Kiss have been disseminated through high-impact scientific journals and presentations at leading international conferences.
The project actively engages in scientific outreach and maintains collaborations to maximise the reach and impact of its findings.
📑Hypothalamic lipidomics and lipid‑sensing pathways in pubertal control
In relation to WP1 and 2, we have published the research article: Central lipid sensing pathways contribute to the control of puberty and its alterations in conditions of obesity. Rodríguez-Vázquez E, et al. Tena-Sempere M (last author). Am J Physiol Endocrinol Metab. 2025 May 1;328(5):E675-E694. This study contains the first mapping of the hypothalamic lipidomic landscape during female pubertal maturation and in conditions of early obesity, and delineates the putative roles of FFAR-, PPAR- and TGR5-related signalling pathways in the control of normal puberty and its alterations in obesity. Related with achievements described in points 1.a. and 2.a.
View more
📑Hypothalamic regulation of puberty and its metabolic control
In relation to WP1 and 2, we have recently published an invited authoritative review, entitled: Hypothalamic control of puberty: from neuronal circuits to mechanisms for its metabolic regulation. Jimenez-Puyer M, Sobrino V, Colledge WH, Jones S, Tena-Sempere M. Reviews in Endocrinology and Metabolic Disorders 2025; doi:10.1007/s11154-025-10001-w. In this review, we summarize some of the recent findings, including those emerging from DOPA-Kiss, on the molecular mechanisms for the metabolic control of puberty and its alterations in obesity.
View more
📑Astrocytic kisspeptin signaling shaping the reproductive axis
In relation to WP2 and 3, we have published the research article: Kisspeptin signalling in astrocytes modulates the reproductive axis. Torres E, et al. Tena-Sempere M (corresponding and lead author), Romero-Ruiz A. Journal of Clinical Investigation 2024;134:e172908. While this study was initiated before the start of DOPA-Kiss, activities conducted in the context of this project permitted the identification of the putative role of kisspeptin signalling in astrocytes as contributing mechanism for pubertal acceleration in conditions of early obesity (see achievements in point 2.f.).
View more
📑Early‑onset obesity and puberty: mechanisms to consequences
In addition, M. Tena-Sempere as PI of DOPA-Kiss was invited as plenary lecturer in the European Congress of Obesity (ECO) 2025, with the conference “Early-onset Obesity and Puberty: From mechanisms to Consequences”, where initial results from the project were presented and discussed in one of the top international conferences in the domain of Obesity. Total number of attendees registered for this conference was over 3000, from Europe and other areas worldwide.
Impact and Future Directions
These findings outline molecular pathways linking metabolic status to pubertal timing and provide a basis for sex‑aware, translational research in obesity‑related pubertal disorders. Next steps prioritize in vivo validation of lipid and nutrient‑sensing routes and the integration of multi‑omics with genome editing to define actionable targets.