Dissecting the Brain Basis of Obesity-Induced Pubertal Alterations: A View to a Kiss (DOPA-Kiss)
Decoding the Role of Kisspeptin Neurons in Puberty and Metabolism
Centered on hypothalamic Kiss1 neurons, DOPA‑Kiss dissects the mechanisms linking nutritional status to pubertal control, with a specific emphasis on early‑onset obesity and sex differences.
Our Mission & Goals
The main mission of DOPA-Kiss is to unravel how kisspeptin neuronal networks integrate metabolic and hormonal signals to regulate puberty onset and reproductive function.
Key goals include:
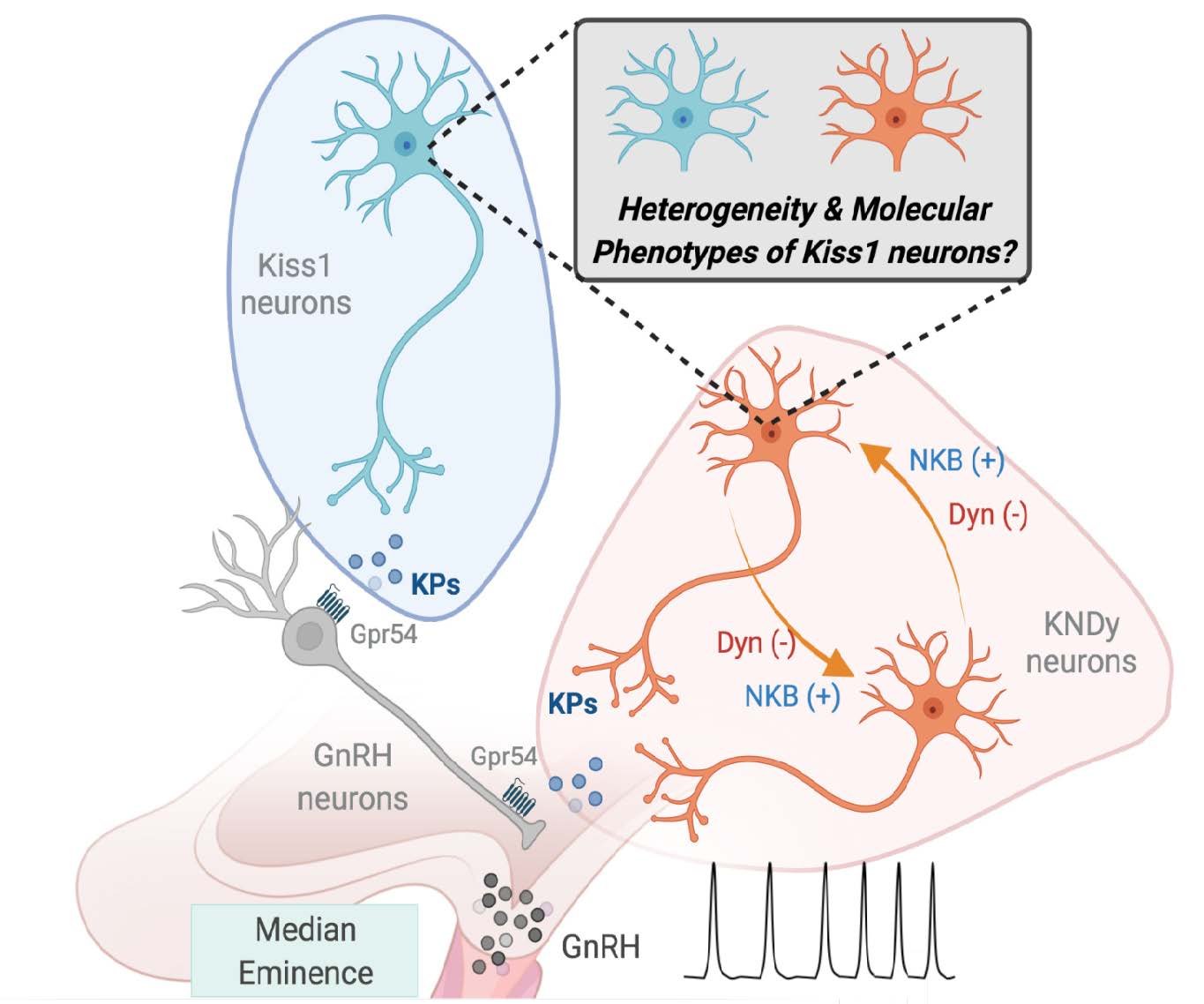
✅ Characterizing the molecular diversity of kisspeptin neurons via integrative multi‑omics across pubertal maturation and early‑onset obesity.
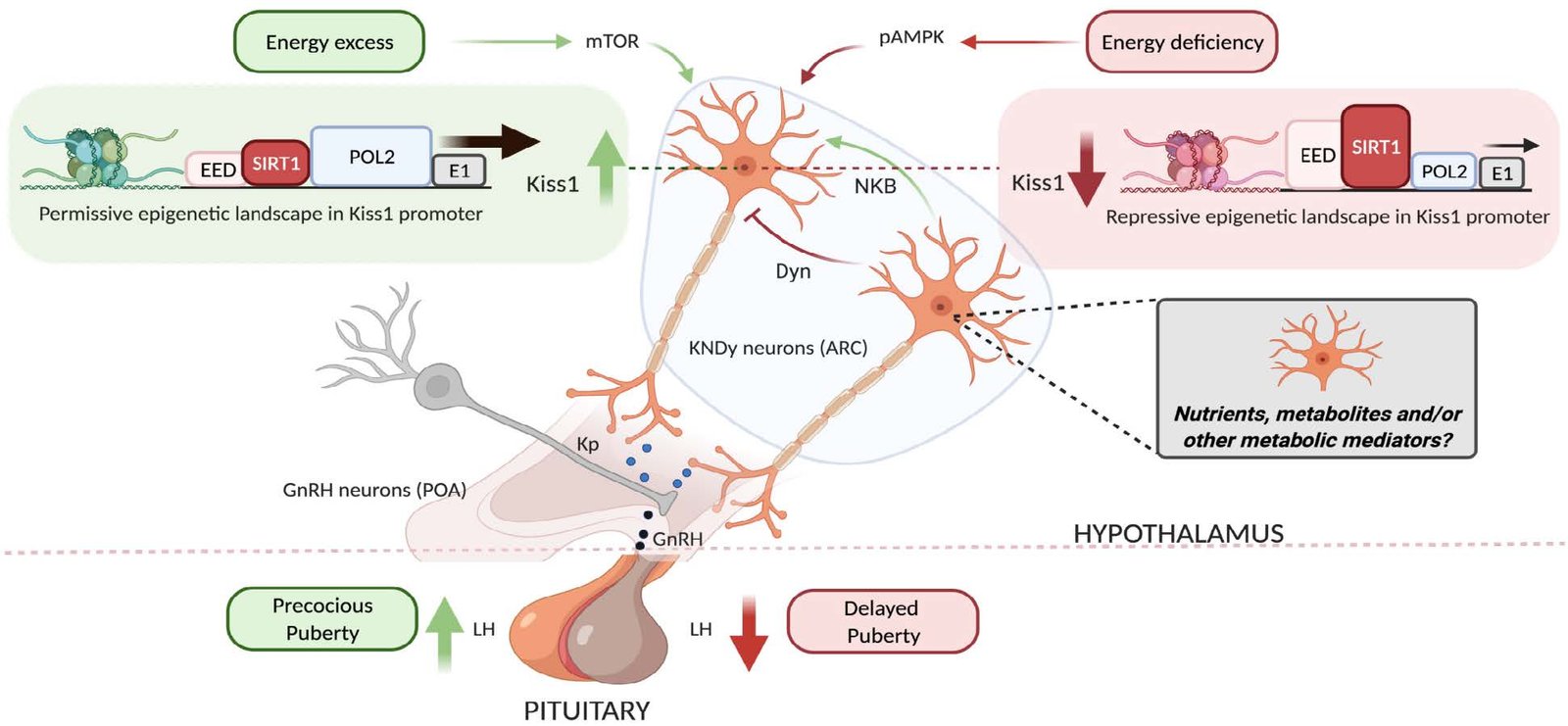
✅ Defining nutrient and metabolic‑sensing pathways (e.g., FFAR/PPAR/TGR5; AMPK/mTOR/SIRT1) that couple energy status to pubertal control.
✅ Establishing causal roles through Cre/LoxP and CRISPR‑based genome editing targeted to Kiss1 neurons in vivo, assessing male–female differences.
✅ Translating mechanistic findings toward a better understanding of human pubertal disorders linked to obesity.
Our Work Packages
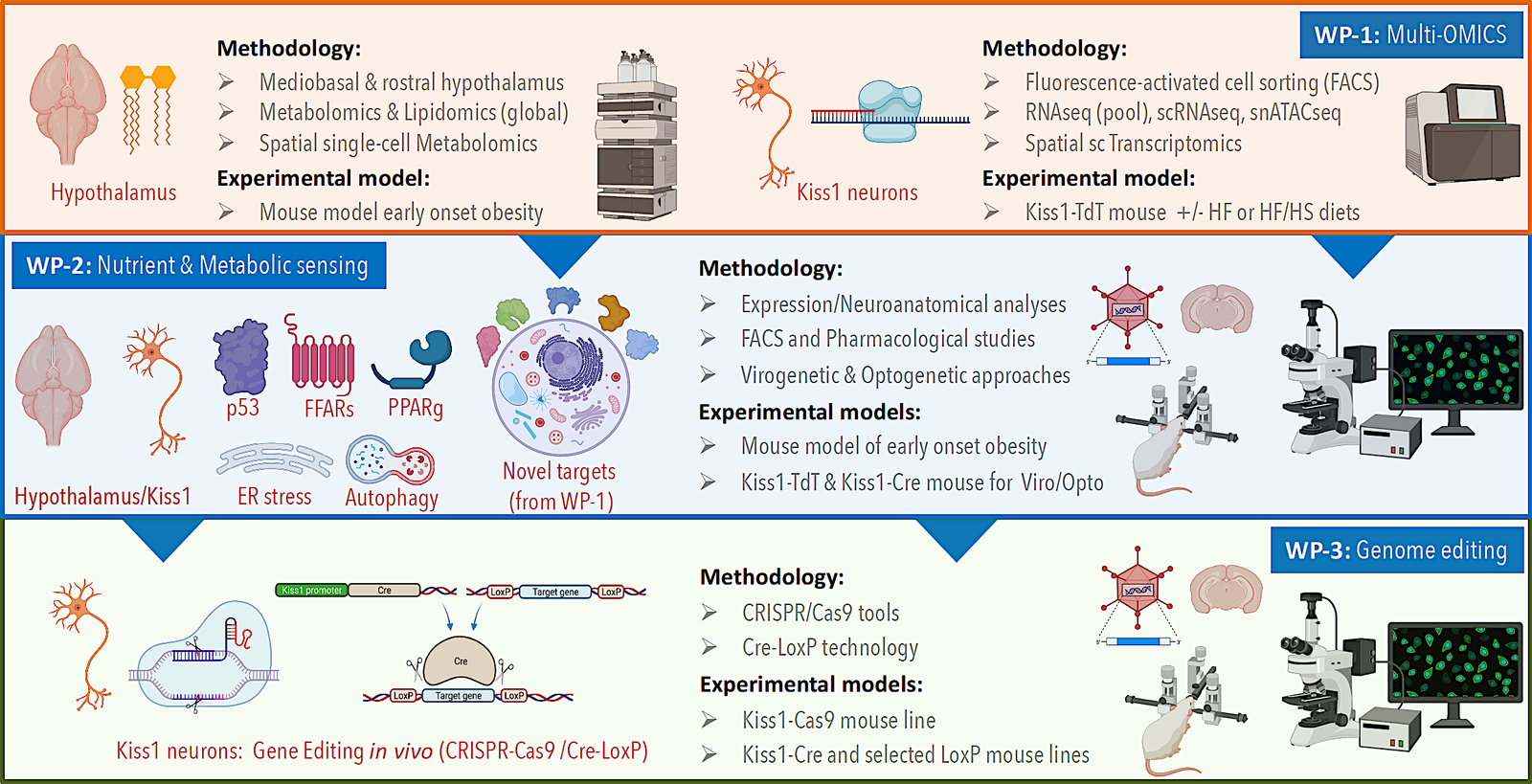
The project is organized into three interconnected work packages, each addressing specific scientific challenges with complementary methodologies.WP1 – Multi-OMICS
Large‑scale molecular profiling of hypothalamus and Kiss1 neuronal populations across pubertal development and in early‑onset obesity.
WP2 – Nutrient & Metabolic Sensing
Mapping lipid and nutrient signaling that modulates hypothalamic circuits and Kiss1 function, including FFAR/PPAR/TGR5‑related pathways.
WP3 – Genome Editing
Applying Cre/LoxP and CRISPR‑Cas tools in vivo to test causal roles of candidate pathways in Kiss1 neurons, with sex‑specific analyses.
Meet Our Team
Led by Prof. Manuel Tena‑Sempere (University of Córdoba/IMIBIC), DOPA‑Kiss brings together expertise in neuroendocrinology, metabolism, and functional genomics to interrogate Kiss1‑centered mechanisms of pubertal control.

Recent Highlights
DOPA‑Kiss is making significant progress in understanding how hypothalamic Kiss1 neurons and metabolic pathways regulate pubertal timing, particularly in the context of early‑onset obesity.
New insights into kisspeptin neuron subtypes
Identification of distinct molecular profiles shaping neuronal function.
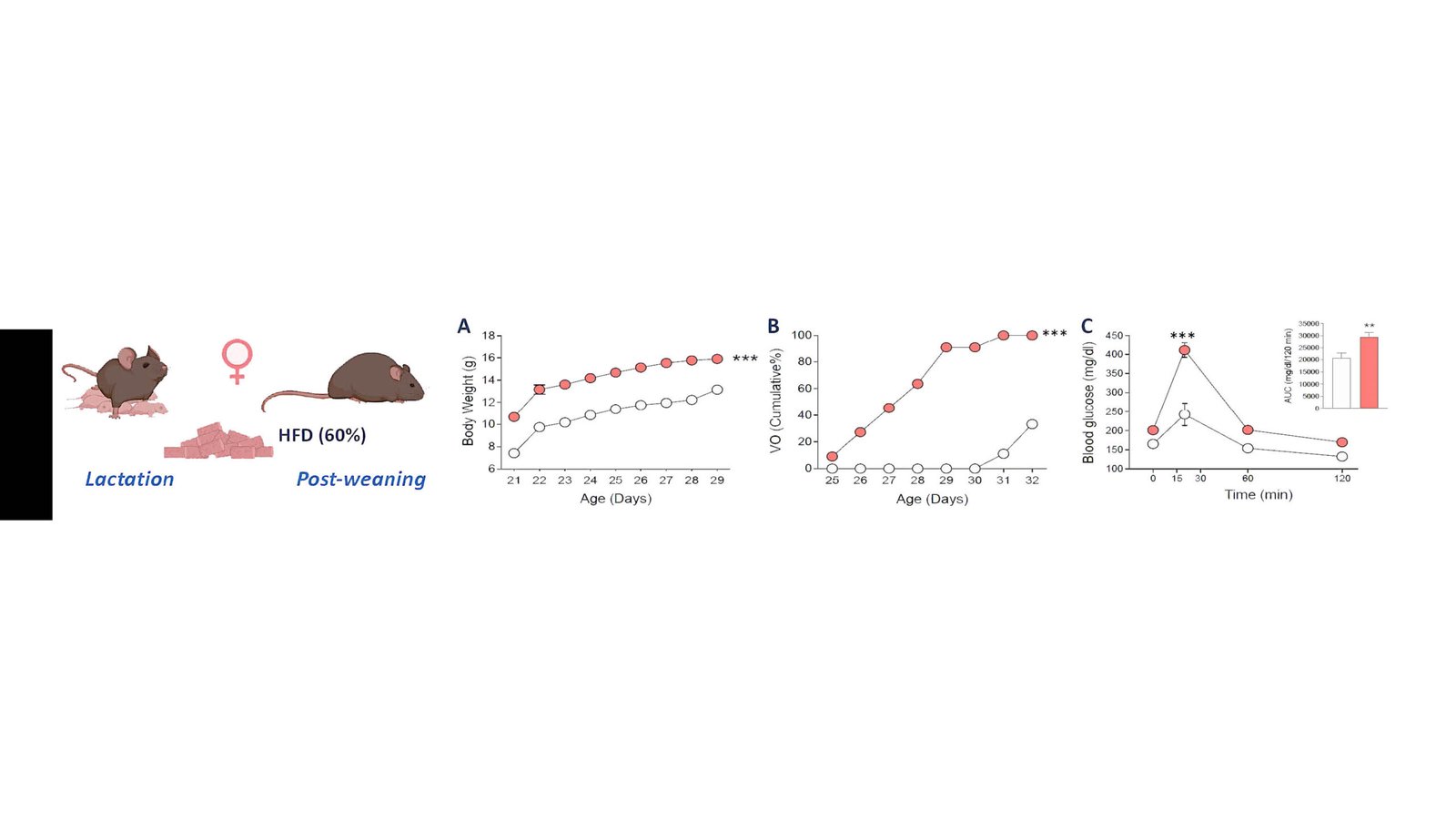
Metabolic regulation of puberty
Evidence on how nutrition modulates hypothalamic circuits.
Advanced CRISPR/Cas9 models
Development of genome-edited models to study puberty onset.
Other Activities
Beyond research, DOPA‑Kiss engages in dissemination, training, and collaboration to accelerate knowledge exchange on obesity‑induced pubertal alterations.


Data Management
Committed to open science and robust data stewardship, DOPA‑Kiss implements a data policy to ensure secure storage, traceability, accessibility, and integrity of project data.